Essential elements for informed consent
The procedure has two main areas that could be problematic when applied in data sharing:
- it is difficult to explain -omics in simple language
- it can be difficult, if not impossible, to provide information about all the potential uses of such data or who might have access to it given the rapid development of both science and technology.
Informed consent should also offer the clear option for the participant to withdraw from the research at any time, which can be challenging as well.
According to the new EU General Data Protection Regulation (GDPR), consent must be freely given, specific, informed and unambiguous.
>For projects linking data to RD-Connect, we recommend that the original informed consent documents contain the following essential elements:
Essential elements for the informed consent of biobanks and observational studies:
- General (name of the PI, Institution, funding, duration, oversight, contact persons)
- Aims, research uses of data (e.g cancer research, RD research)
- Voluntariness of participation and possibility to withdraw
- Procedures involved in participation, including interviews, blood taking, etc.
- Kinds of samples and data that will be collected
- Potential physical, psychological and social risks
- Potential benefits
- Description of the coding system
- Protections in place locally to ensure the confidentiality of samples and data
- Access to data/samples for research purposes: who will have access who should control and what the procedures in place (data access committee)
- Access to data/samples for purposes such as validation, quality control, etc.
- Study oversight
- Compensation/reimbursement
- Custodianship of samples
- Study dissemination
Core elements for the informed consent of studies participating to RD-Connect:
- Hosting of the data in an open access database
- Access by industry if foreseen
- Possible linkage to different databases (patient registries, medical records, etc.)
- Possibility of large scale genome sequencing techniques
- Possibility of data sharing across research groups and national borders
- Return of incidental findings
- Withdrawal procedures, such as sample retrieval and/or destruction
- Prospects for third-party commercialization and intellectual property procedures;

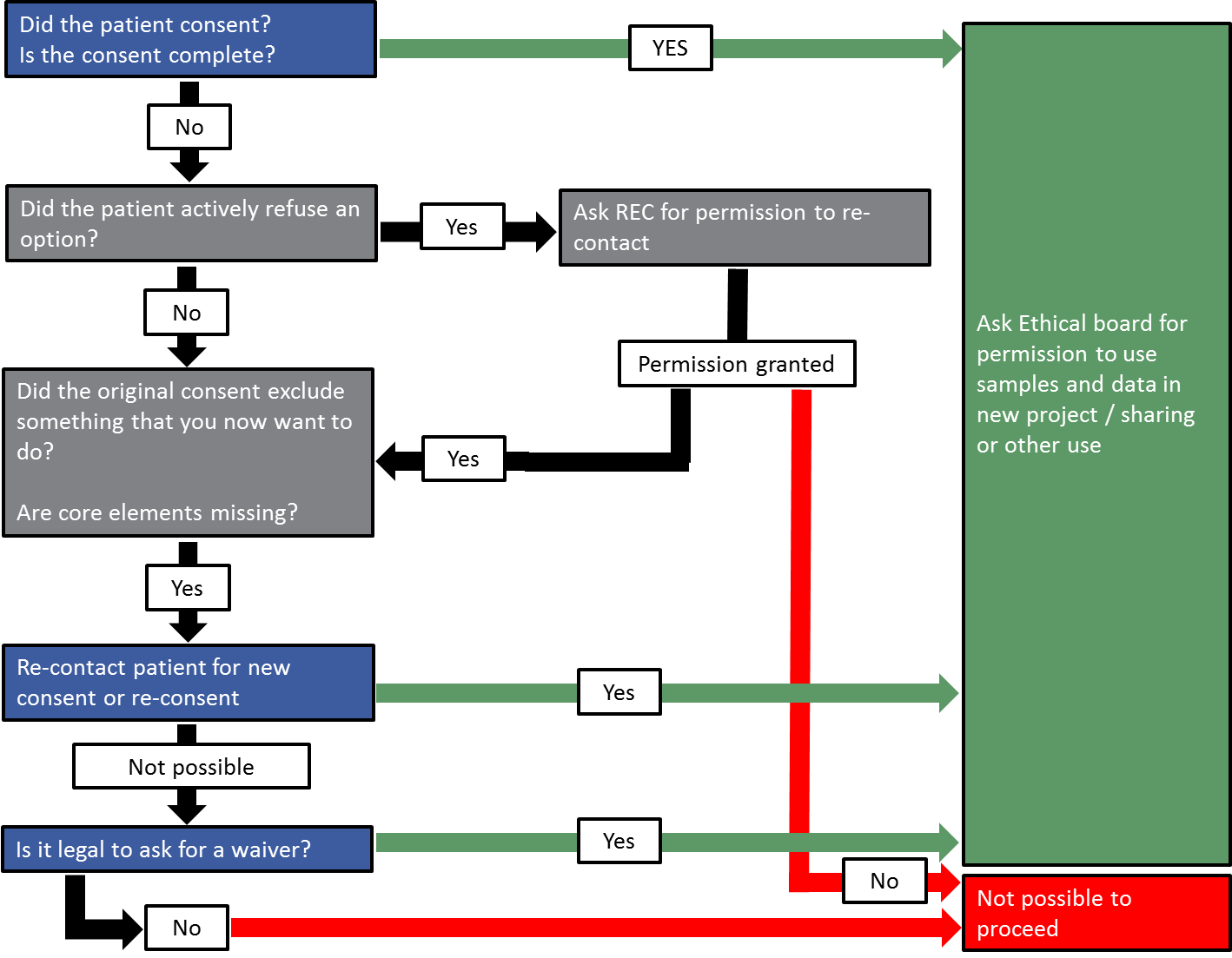
Retrospective consent
One issue faced by rare disease research projects is using ‘historically collected’ biomaterial samples – those collected from patients before the project began.
In some cases, the consent given by the patients did not include permission for data/sample sharing or the permission for omics research and genetic sequencing. This was often because these uses were simply not predicted at the time the sample was taken.
Data and sample sharing as well as -omics technologies are now key to rare disease research and RD-Connect’s objectives. With this in mind, the Rare Disease Patient and Ethics Council (RD-PEC) and Patient Advisory Council (PAC), in close collaboration with the partner research projects NeurOmics and EURenOmics, and with the wider rare disease patient community, established recommendations to ensure that as many as possible of these precious historic sample collections may be used. Consultation with patient groups and researchers using the samples has highlighted the determination to find ways to overcome barriers put in place by out-of-date or incomplete consent. As a result, the RD-PEC and PAC have drawn up a guideline procedure for using already collected (historic) samples.