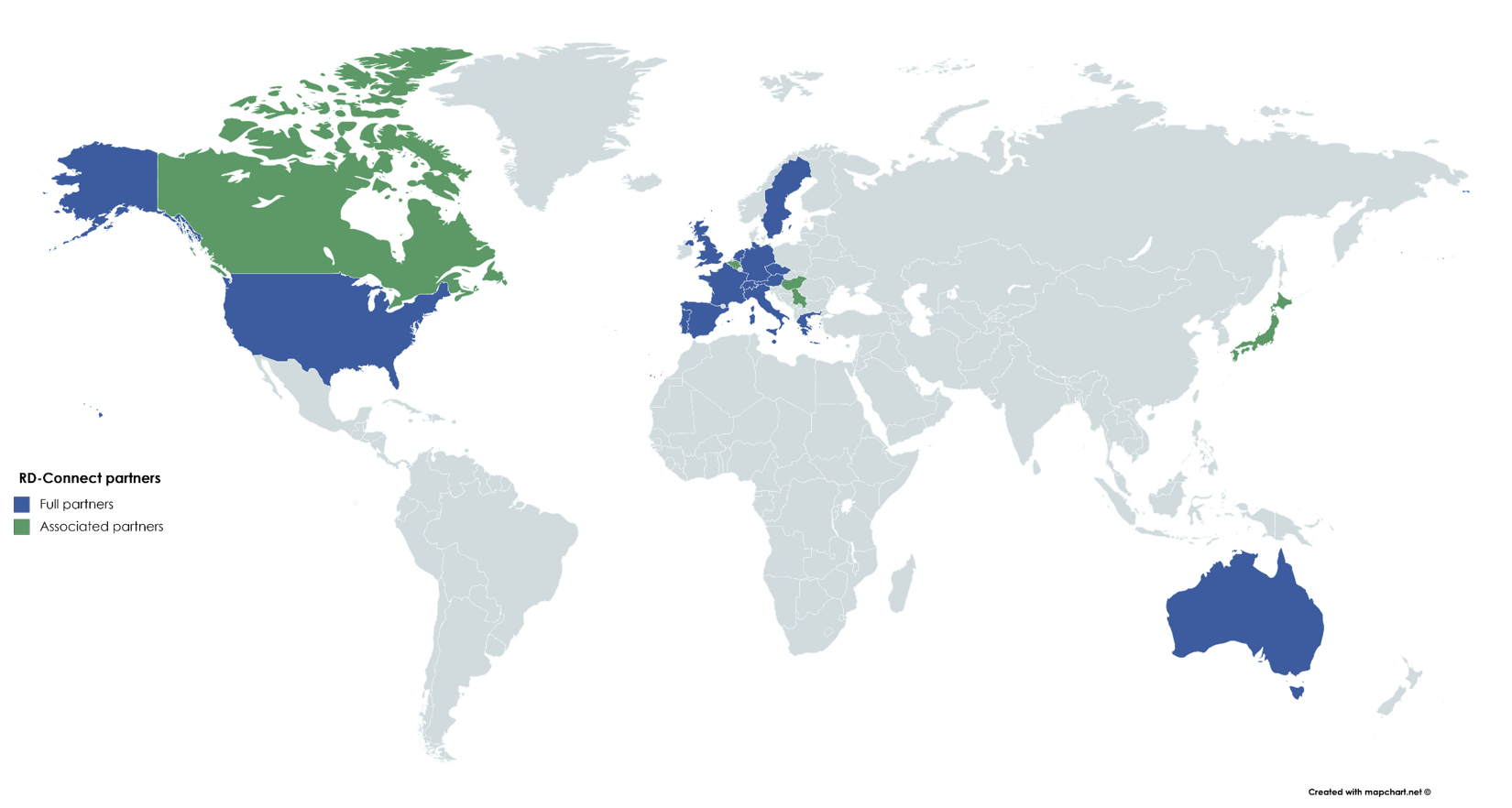
The RD-Connect project, which gave rise to the RD-Connect Community, was a collaborative work of a number of partners working on eight different areas (work packages) of developing the rare disease research infrastructure. RD-Connect worked across the EU and beyond, with different working teams based in different countries.
Coordination and Management
The RD-Connect coordination office, based at the Newcastle University in the UK, was led by Hanns Lochmüller (2012-2017) and managed by Emma Heslop (2012-2016) and Libby Wood (2017-2018). The coordination team led on the strategic direction of the project, along with being responsible for financial and communication activities.
Patient registries
The work on linking up rare disease patient registries across the world, led by Domenica Taruscio, was conducted by teams at the Istituto Superiore di Sanità in Rome, Italy and Medical University of Graz, Austria. They have developed Registry & Biobank Finder, a global directory of patient registries and biobank for rare disease research. To support data exchange, the team has developed standards of data interoperability for patient registries. Read more>>
Biobanks
The biobank team at the Fondazione Telethon in Milan, Italy, led by Lucia Monaco, and University Medical Center Groningen, the Netherlands, focused on connecting biobanks and developing the RD-Connect Sample Catalogue, which allows researchers browse individual biosamples. They worked closely with the patient registry team, to integrate information on the existing biobanks and patient registries in the common Registry & Biobank Finder. Read more>>
Data linkage
The RD-Connect partners in the Netherlands (Leiden University Medical Center and University Medical Center Groningen) focused on the technical aspect of linking different types of data between institutions across countries. The Data Linkage Plan worked on implementing the FAIR (Findable, Accessible, Interoperable, Reusable) principles to allow queries and analyses on different data sources without the requirement to bring these data physically together. The team collaborated closely with the patient registry, biobanks and Genome-Phenome Analysis Platform teams. Read more >>
Bioinformatic tools
To enable novel types of analysis of next-generation sequencing data, several bioinformatics teams working in RD-Connect, developed a number of innovative bioinformatics tools. Read more>>
Genomne-Phenome Analysis Platform
The team led by Ivo Gut at the Centro Nacional de Análisis Genómico in Barcelona, Spain, developed the Genome-Phenome Analysis Platform that enables the users to analyse next-generation sequencing data and connect them to clinical information. The work was coordinated by Sergi Beltran Agulló. Read more>>
Ethical, Legal and Social Issues (ELSI)
To ensure protection patient privacy without compromising research and drug discovery, the team of ethicists and lawyers led by Mats Hansson at the Centrum För Forsknings- & Bioetik of the Uppsala University, Sweden, provided guidance to researchers and work on the most appropriate models for data sharing. The ELSI work also included the interdisciplinary Rare Disease Patients Ethics Council. Read more>>
Science Communication and Impact
Ensuring that the work of RD-Connect has an impact on research, diagnosis and the quality of life of patients is the focus of the Impact team, lead by Kate Bushby at the Newcastle University, UK. The work on RD-Connect monthly newsletters, website, media and other dissemination channels is coordinated by Dorota Badowska.
Patient engagement
Patient engagement in RD-Connect is coordinated by Virginie Bros-Facer from the European Rare Diseases Organisation EURORDIS. The Patient Advisory Council informs the project partners of issues important to patients, which allows ensuring that all project activities have a patient-centric approach throughout. Read more >>